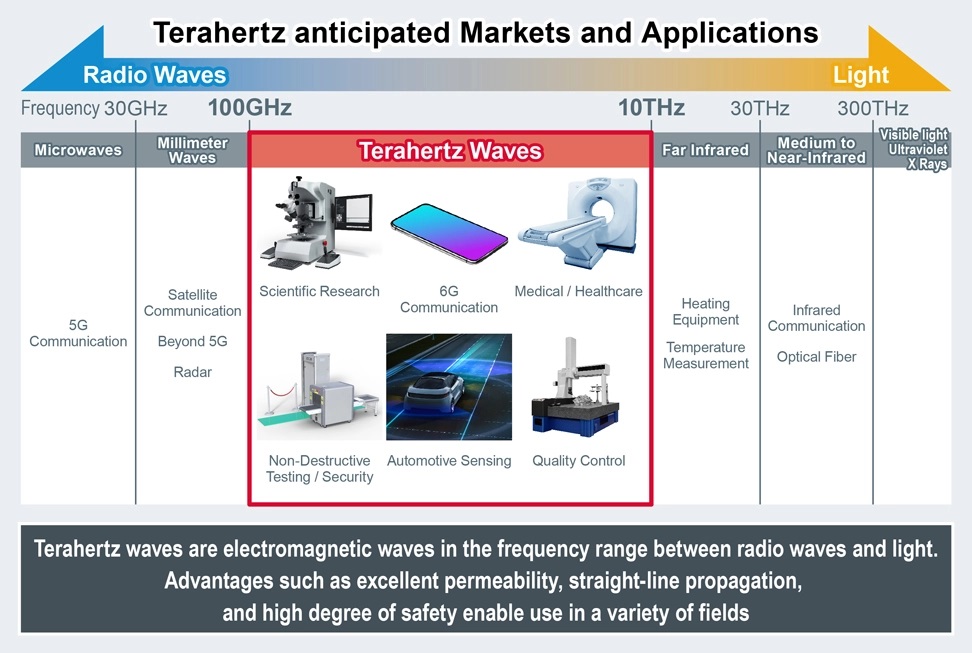
SANTA CLARA, CA and KYOTO, Japan, Feb 3, 2025 – ROHM Semiconductor has announced that they have started offering samples of the industry’s smallest terahertz (THz) wave oscillation and detection devices utilizing semiconductor elements known as Resonant Tunneling Diodes (RTDs). Terahertz waves are anticipated to be applied to non-destructive testing, imaging, and sensing in the medical and healthcare sectors, as well as potentially future ultra-fast communication technologies. Providing these devices contributes to the advancement of THz wave applications.
ROHM has developed a 0.5mm × 0.5mm RTD chip for terahertz wave generation and detection, capable of oscillating and detecting terahertz waves at a frequency of 320GHz (typ.) with an output power ranging from 10 to 20µW. ROHM will begin offering samples of this RTD element mounted in a PLCC package (4.0mm × 4.3mm) commonly used for LEDs. With a compact size of one-thousandth that of conventional oscillators, the innovation enables development of terahertz wave applications, including in space-constrained environments.
By positioning the antenna surfaces of the oscillation and detection devices facing each other 10mm apart, a dynamic range of 40dB (typ.) can be achieved. Both oscillator and detector maintain a drive power consumption of 10mW (typ.), while their ability to oscillate and detect terahertz waves at room temperature eliminates the need for cooling equipment required with some conventional methods. These power-saving devices are unaffected by the operating environment, enabling use in a wide range of applications.
ROHM offers samples of terahertz wave oscillation and detection devices at a sample price of $990.0/unit (excluding tax), which is less than one-tenth the price of conventional devices. ROHM also provides evaluation kits that include an evaluation board and other components, allowing users to easily integrate the devices into a research and development environment.
Ken Nakahara, general manager of center, ROHM R&D Center, ROHM Co., Ltd. said, “The terahertz (THz) R&D team and I are very excited and proud to have reached the point where we can bring this technology to market. We have dedicated ourselves to THz devices for about 15 years; the journey has not been easy. We gathered the forces of industry, academia, and government along the way, and have successfully established the position that ROHM holds today. The team understands that this announcement is just a small step toward the commercialization of THz technology, but at the same time, it is a giant leap for us. We believe that this small beginning will grow big and contribute to the well-being of our society.”
About ROHM
ROHM Co., Ltd., established in 1958, is a Japanese electronics manufacturer headquartered in Kyoto, Japan. The company specializes in designing and producing a wide range of electronic components, including integrated circuits (ICs), semiconductors, and optoelectronics. ROHM serves various industries such as automotive, industrial equipment, consumer electronics, and telecommunications. Their product portfolio encompasses monolithic ICs, power modules, transistors, diodes, light-emitting diodes (LEDs), and capacitors. As of 2017, ROHM reported annual revenues of approximately ¥352 billion. With a global presence, the company operates in regions including Asia, the Americas, and Europe, supplying high-quality electronic components to clients worldwide.
Source: ROHM



